General information
Scientific Name
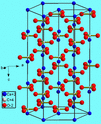
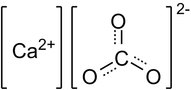
The scientific name of chalk is Calcium Carbonate.
Chemical Formula
The chemical formula for Chalk (Calcium Carbonate) is CaCO3
Other name/names for Calcium Carbonate
The only other common name for Calcium Carbonate is Chalk which is known in everyday schools and in the everyday world.
The only other common name for Calcium Carbonate is Chalk which is known in everyday schools and in the everyday world.
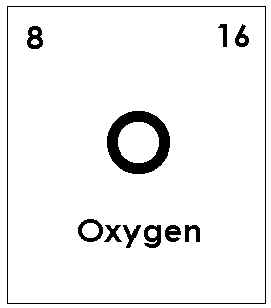
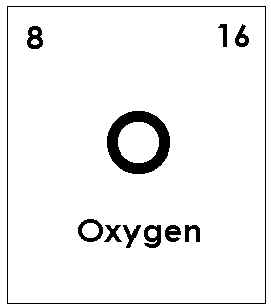
Number of atoms in each element of Calcium Carbonate
Number of atoms of:
Calcium: 1
Carbon: 1
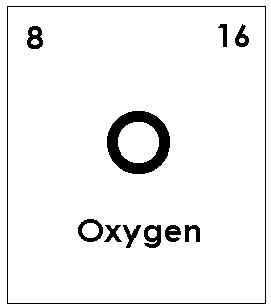
Oxygen: 3
Total: 5
Calcium: 1
Carbon: 1
Oxygen: 3
Total: 5
|
Physical Properties of Calcium Carbonate
Chalk is usually white in color and it is easily breakable. It is not malleable because if a person hammers it, it will simply break. Thus, chalk is also not ductile. It is porous, which means that it has tiny holes through which air water can pass through. Chalk is not smooth, instead, it is very rough in texture. These physical properties also show that chalk is a non-metal.
|
Chemical Properties of Calcium Carbonate
The chemical properties of chalk are the following. Chalk is already a molecule which means that different elements have reacted, and their valance electrons have moved or shifted to complete their valance shells. This makes chalk not very reactive. Chalk is not combustible. It does not allow electrons to pass through which makes it not a very good conductor of electricity.
|